Contact person: Dr. Anders Lundgren and Prof. Dr. Erik Reimhult
We investigate the colloidal interactions of bacteria with focus on early stages of attachments of bacteria to surfaces. Although biological systems are usually understood through the lens of specific biological interactions our interest is the ubiquitous nonspecific colloidal interactions that are present in all and dominate most, also biological, systems. Specific interactions are only short-range (Ångström) while non-specific interactions span tens of nm. Bacteria thrive in a variety of environments and it is therefore plausible that their ability to manage non-specific interactions are instrumental to their abilities.
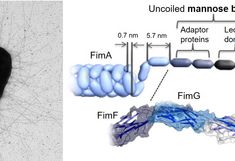
An important example of the ability of bacteria to adhere to many surfaces as well as an important predictor of their virulence is the existence of non-sexual pili or fimbriae extending from the cell membrane. Many bacteria interact with surfaces via long (µm) and thin (nm) tethers through these fimbriae. For Escherichia coli (E. coli), type I fimbriae are implicated in the formation of pathogenic biofilms in the urinary tract via both specific and non-specific bonds. Colonization of abiotic surfaces such as catheters using nonspecific bonds have severe impact on hospital care and human suffering. We investigate the role of fimbriae in the interaction of bacteria under shear with surfaces displaying different densities of nanoscopic mannose or hydrophobic domains, produced by functionalization of self-assembled nanoparticles on PEGylated glass substrates. The tip of type I fimbriae form specific bonds with domains of mannose, implicated in so-called catch bonds, while only non-specific bonds can form to hydrophobic patches of the same dimensions, raising the interesting question of what advantage the fimbriae provide a bacterium with on abiotic surfaces.
We investigate this question using a combination of nanopatterned surfaces, microfluidics, traditional biofilm cultivation techniques and new developments in digital phase contrast holography.