Kathrin Göritzer
Glyco-Immunology

Research Interests
Orientation
Our lab investigates how protein glycosylation shapes antibody assembly, stability, and function, and translates these mechanisms into design rules for next‑generation biologics, with a focus on effective mucosal and topical delivery.
- Decode glycan‑mediated effects on antibody interactions with microbes, host receptors, and barrier environments (mucosal, epithelial, cutaneous).
- Define how N‑ and O‑glycosylation influence effector functions, Fc receptor engagement, pharmacokinetics, and protease resistance.
- Engineer glycosylation and folding/quality‑control pathways to steer antibody assembly, stability, and manufacturability.
- Optimize formulations and delivery for topical use, including stability in secretions, muco‑/bioadhesion, residence time, and microbiome compatibility.
- Advance plant‑based biomanufacturing (Nicotiana benthamiana) for high‑quality recombinant antibodies and complex multimeric formats.
- Translate structure–function insights into candidates for prophylaxis and therapy across infectious disease, inflammation, and oncology.
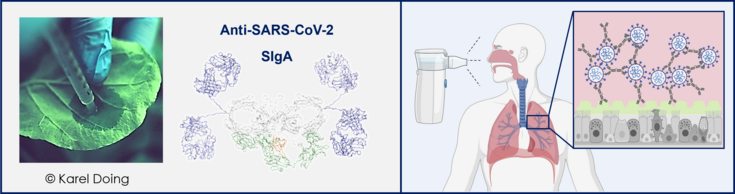
Composite graphic illustrating plant-based production and mucosal delivery of anti–SARS‑CoV‑2 antibodies: left, syringe infiltration of a Nicotiana leaf; center, structural model of dimeric secretory IgA; right, a nebulizer delivering antibody to the respiratory tract, with inset showing SIgA cross‑linking viruses in mucus above airway epithelium.
Approach: in planta glyco‑engineering, ER folding/chaperone modulation, glycomics and proteomics, biophysical analyses, and functional cell-based assays.
Current Projects
Structure–Function Relationships of Human SIgA Glycosylation
SIgA’s superior neutralization, anti-inflammatory properties, and stability in secretions are closely linked to its extensive N‑ and O‑glycosylation. However, tissue-resolved SIgA glycosylation profiles and their functional significance remain insufficiently characterized.
We combine tissue-informed glycan mapping with in plant glyco‑engineering of human glycoproteins to generate fully assembled SIgA bearing homogeneous, tailored glycans. Resulting variants are subjected biochemical and biophysical characterization and to in vitro and cell-based assays to establish causal links between glycan identity and function.
The ability to produce SIgA with controlled, homogeneous glycoforms utilizing the innovative plant-glycoengineering platform overcomes heterogeneity in conventional systems, enabling precise dissection of glycan contributions to SIgA biology and informing the development of robust mucosal antibody therapeutics.
Publications
Funding Agencies
Collaborations
Julian Ma, City and St. George’s, University of London, UK


