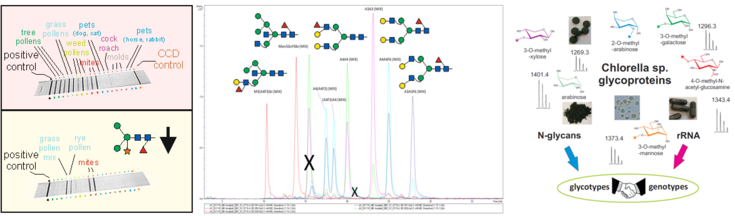
Glycoprotein analysis
Oligosaccharides on secreted proteins are highly complex and diverse structures, which differ by the number and position/linkage of several different sugar residues. We develop methods for a true isomer-sensible glycan analysis using our modern LC-MS equipment to the best. Reference glycans are synthetized using recombinant glycosyltransferases. Our curiosity leads us into the kingdoms of plants, insects, algae and fungi. Most efforts are however dedicated to pharmaceutically relevant glycoproteins.
A particular focus is on the development of appropriate methods for mining the enormous piles of data provided by LC-MS analysis in the field of glycoproteomics and glycomics. The self-developed programs can be found in the "Stadlmann Lab".