Molecular Mechanisms in Plant Adaptation
Overview
Degradation of plasma membrane proteins, ESCRT, Ubiquitin, Intracellular transport of proteins
Leader: Priv.Doz. Mag. Dr. Barbara Korbei
Orientation
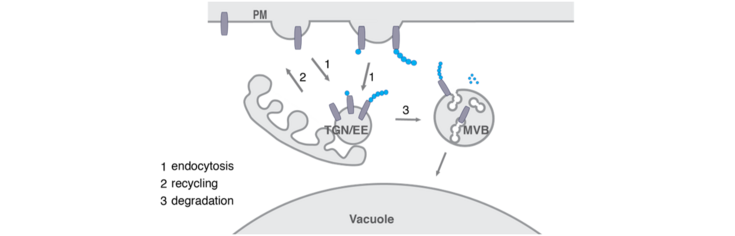
Due to accelerating climate change and the resulting rapid increase in extreme environmental conditions, the question of how plants can flexibly adapt to their constantly changing environment is a highly topical area of research. Plants, being sessile organisms, have evolved a plethora of mechanisms to be able to respond quickly and accurately to their surroundings. The plasma membrane serves as boundary between the inside and the outside of the cell and is densely packed with proteins that are responsible for sensing and transmitting stimuli. Plants thus have to be able to rapidly and accurately adjust not only the localization but also the amount of their plasma membrane proteins. This is organized by endosomal trafficking, which transports membrane proteins to and from their site of action. Here, the post-translational modification of plasma membrane proteins with the small protein ubiquitin is key in signaling their removal from the plasma membrane by endocytosis and further degradation in the vacuole. To understand the significance and the variety of roles played by this reversible modification, the function of ubiquitin receptors, which translate the ubiquitin signature into a cellular response, needs to be elucidated.
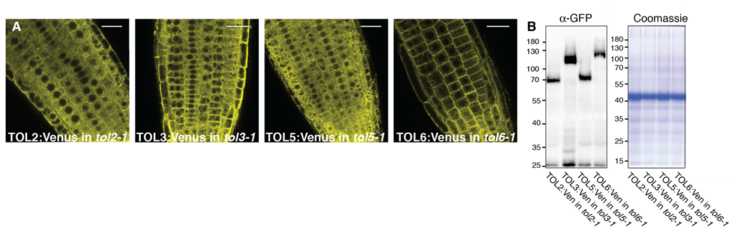
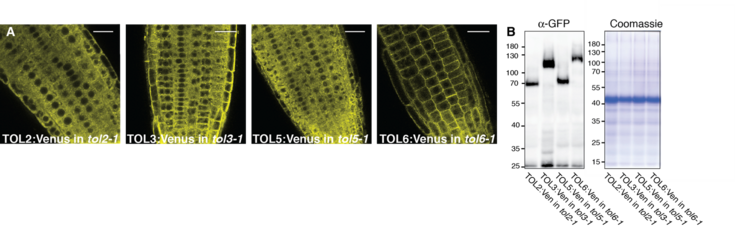
In this lab we try to unravel the function of the TOM1-like (TOL) protein family in the recognition and in initiating sorting to the vacuole of ubiquitinated plasma membrane proteins destined for degradation. The TOL proteins function in plants as multivalent ubiquitin receptors, governing ubiquitinated cargo delivery to the vacuole via the conserved Endosomal Sorting Complex Required for Transport (ESCRT) pathway.
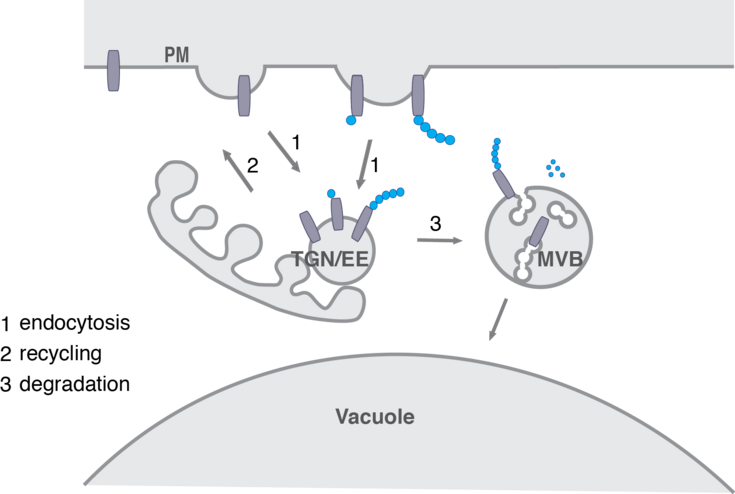
For this purpose, we conduct in-depth functional characterization of the TOL protein family in the endosomal system of the model plant Arabidopsis thaliana and asses their binding to conserved elements of the endosomal pathways. This helped to intensify functional links between TOL proteins and the plant sorting machinery. The work from our lab will contribute to clarifying how the TOL proteins fulfill their function in acting as principal gatekeepers in the degradation of ubiquitinated plasma membrane proteins with a special focus on their ubiquitin binding activity and their regulation by ubiquitination.
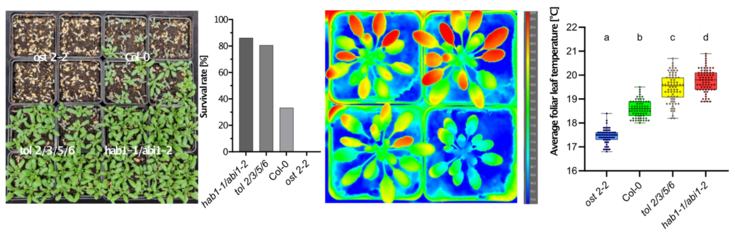
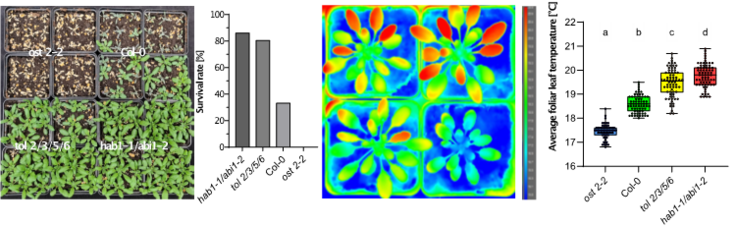
Furthermore, responses to stresses, like drought stress, are regulated and coordinated by phytohormones, whose transport and perception are commonly centered around plasma membrane-localized proteins. We are currently working on assessing the role of specific TOL proteins in the abscisic acid (ABA) signaling pathway, where we found a higher order tol mutant plant line, which is less sensitive to drought stress and hypersensitive to the plant hormone ABA. This indicates that the TOLs, albeit apparent redundancies, play a role in very specific pathways where they may be relevant for fine-tuning of plant hormone signaling by means of tightly controlled protein turnover. Understanding the function of TOLs could give key insights into mechanisms by which plants define the trade-off between stress tolerance and plant development when faced with challenging environments. Findings obtained in the model plant Arabidopsis thaliana provide a solid foundation for translational research aimed at breeding more tolerant crops.