Modulation of the high ambient temperature responsevia endosomal trafficking in Arabidopsis thaliana
SUPERVISOR: Barbara KORBEI
PROJECT ASSIGNED TO: Nils LEIBROCK
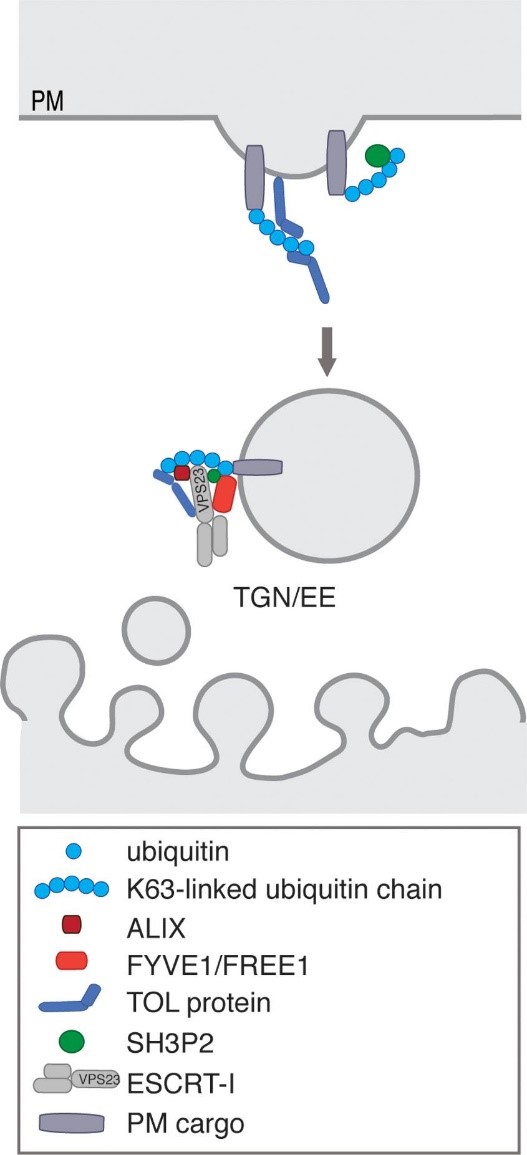
Plants are sessile organisms that have developed various strategies to adapt to their ever-changing environmental conditions. Especially in times of climate-change, their fundamental strategies need to be further explored to offer innovative solutions. One of these strategies revolves around how plants regulate protein degradation and translocation, where molecular trafficking via the endosomal pathway can help plants maintain cellular homeostasis and assist in stress responses. In the endosomal trafficking pathway, cargo deemed for degradation or translocation can be ubiquitinated. The ESCRT (endosomal sorting complex required for transport) is responsible for recognition of ubiquitinated cargo and sorting it into intraluminal vesicles for degradation in the vacuole. While mammals can rely on the functionality of ESCRT-0 subunits for cargo recognition and sorting, plants only possess the remaining ESCRT-I, -II and -III subunits. In plants, TOL- (TOM1-like) proteins are key in the initial recognition and sorting of ubiquitin-tagged proteins functioning as substitutes for the elusive ESCRT-0. The aim of this study is to prove their fundamental importance in plant physiology and the protein degradation network. Next to their ubiquitin-binding sites, TOLs contain a variety of domains for interactions with other ESCRT- along with other non-ESCRT-subunits for downstream signalling and cargo processing. We have proven the participation of distinct TOL protein family members in the transmission of environmental signals such as gravity, drought stress and ABA signalling. Following these findings, we will use the functional variety of this protein family to unravel their importance in plant responses to high ambient temperature. A double tol mutant plant line exhibiting aberrant growth at high ambient temperature has been identified. With this project we aim to reveal how the distinct phenotype results from altered hormonal crosstalk together with adjustments from other protein degradation pathways such as selective autophagy.