Human Heme Peroxidases
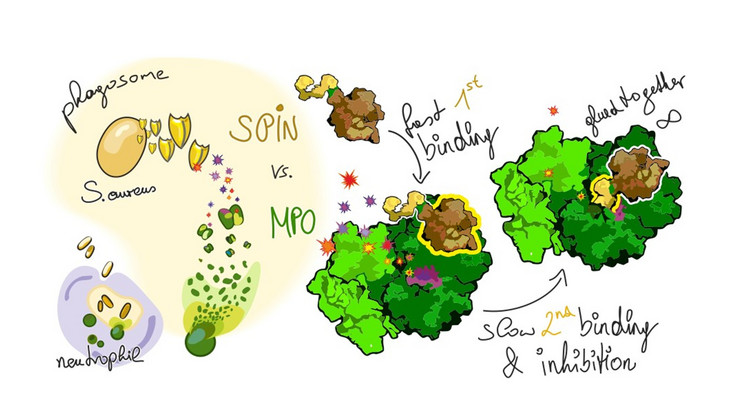
Understanding the structure and function of human heme peroxidases is crucial for both basic biology and biomedical research. This enzyme family—including myeloperoxidase, eosinophil peroxidase, lactoperoxidase, peroxidasin, and thyroid peroxidase—plays key roles in host defense, tissue organization, and hormone biosynthesis. They catalyze reactions that generate reactive oxidants, which help eliminate pathogens but must be tightly regulated to prevent tissue damage.
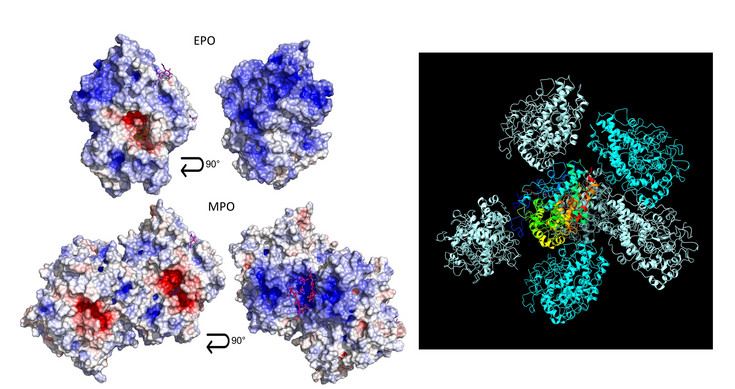
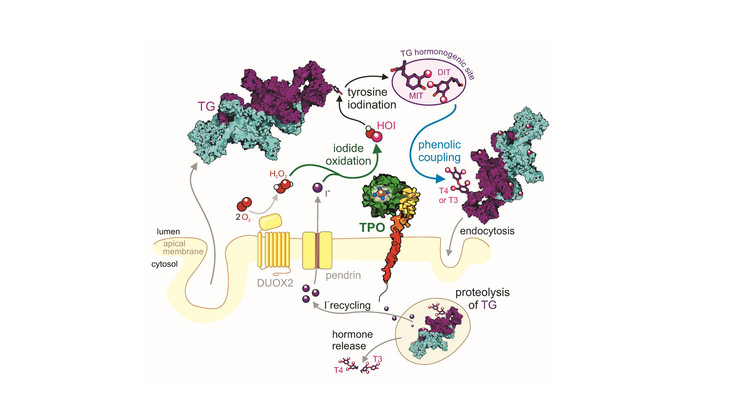
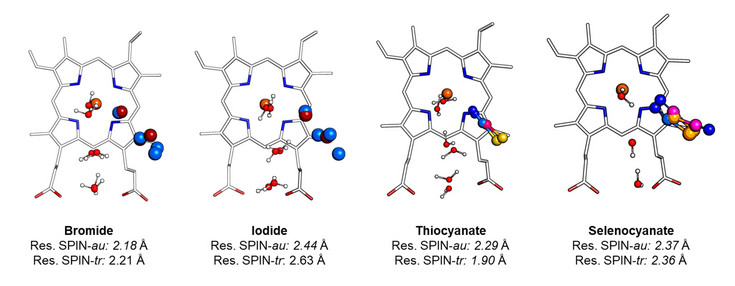
At the core of their activity lies the heme group, an iron-containing cofactor whose surrounding protein structure determines enzyme reactivity, specificity, and stability. Even small structural differences can lead to distinct biological functions—for example, peroxidasin is involved in extracellular matrix formation, while thyroid peroxidase is essential for thyroid hormone production.
Understanding these structure–function relationships is highly relevant to human health. Dysregulation of heme peroxidases has been linked to inflammatory diseases, cardiovascular conditions, and endocrine disorders. By studying how these enzymes work at a molecular level, we can identify mechanisms of disease and develop targeted therapies or diagnostic tools.
In summary, insight into human heme peroxidases connects molecular structure with physiological function and disease, making it a key area of research with broad clinical and biotechnological relevance. To acchieve the necessary fundamental understanding we use a combination of biochemical analysis, X-ray crystallography and molecular biology.