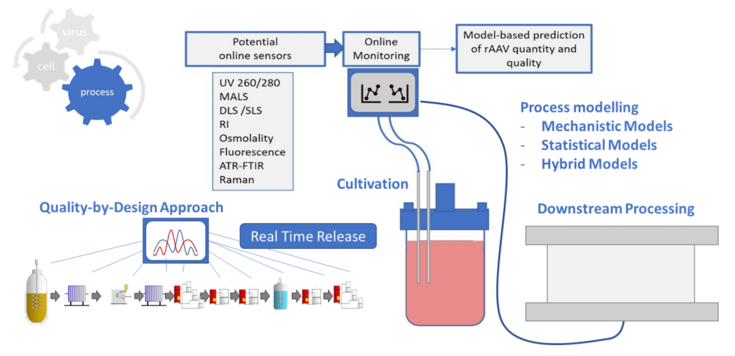
Online Monitoring in Downstream Processing of AAVs
SUPERVISOR: Astrid DÜRAUER
PROJECT ASSIGNED TO: Martina WINTER
Gene therapy offers the opportunity to treat diseases rooted in genetic defects by introducing genetic material into the target cells1. For viral vector-based delivery, recombinant Adeno associated viruses (rAAVs) are frequently used which are mostly produced in mammalian cells such as human embryonic kindney cells (HEK cell lines). Following the initial approvals of treatments based on rAAVs in the last ten years2,3, AAV production must now progress into next-generation manufacturing in order to effectively provide these new medicines.
One significant challenge lies in the cost-intensive nature of rAAV production. Moving towards next-generation manufacturing methods necessitates a shift towards knowledge-based processes, enabling cost-effective and sufficient supply of these biopharmaceuticals. This transition requires a deeper understanding of downstream bioprocessing. Traditional quality assessment methods, reliant on offline analysis, offer only retrospective insights into product quality, resulting in increased costs, extended holding periods, and limited process control during AAV production. Therefore, there is a growing emphasis on developing inline and at-line sensors to establish soft sensors for real-time monitoring systems and predictive models. Such advancements aim to enhance process control both upstream and downstream in the rAAV production process, ultimately improving efficiency and safety.
The aim of this thesis is online monitoring with the goal of model predictive control, for which mechanistic, hybrid and statistical models in chromatographic processes will be established.
1. Dunbar, C. E. et al. Gene therapy comes of age. Science (1979) 359, (2018).
2. El Andari, J. & Grimm, D. Production, Processing, and Characterization of Synthetic AAV Gene Therapy Vectors. Biotechnol J 16, (2021).
3. Wang, D., Tai, P. W. L. & Gao, G. Adeno-associated virus vector as a platform for gene therapy delivery. Nat Rev Drug Discov 18, 358–378 (2019).
