Recombinant protein extraction with Polymers
SUPERVISOR: Rainer HAHN
PROJECT ASSIGNED TO: Alexander JURJEVEC
Industrial applied cell disruption methods like high- pressure homogenization (HPH) lead to full extraction of recombinant proteins from E. coli cells but also to complete destruction of the cell assembly. As a result, large amounts of host cell proteins (HCPs), DNA and endotoxins are also released. These impurities pose a challenge for further downstream purification and have to be completely removed from the final product. [1][2][3].
The aim of this doctoral thesis is to develop a new extraction method that not only leads to complete protein extraction, but also to lower impurity levels.
Polymers are known for their good clarification properties of E. coli components such as residual DNA and endotoxins. On the other hand, they also possess permeabilizing properties of the cell membrane of E. coli. [4][5] [6]. We want to take advantage of these properties and develop a new extraction method which outperforms standard extraction methods like HPH. In addition, a detailed understanding about the extraction mechanism and kinetics should be gained. Results from extraction of cytoplasmatic GFP from E. coli are shown in 1 and not only show complete GFP extraction but also reduced HCPs.
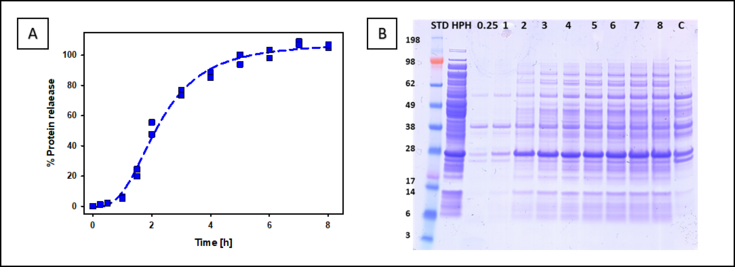
Figure 1: Extraction efficiency of GFP with polymers in comparison to HPH. (A) Extraction kinetic (B) SDS – PAGE of the Extraction kinetic
[1] A. P. J. Middelberg, Microbial Cell Disruption by High-Pressure Homogenization. Humana Press, 2000.
[2] C. Schimek et al., “Extraction of recombinant periplasmic proteins under industrially relevant process conditions: Selectivity and yield strongly depend on protein titer and methodology,” Biotechnol. Prog., vol. 36, no. 5, pp. 1–11, 2020.
[3] B. Balasundaram, S. Harrison, and D. G. Bracewell, “Advances in product release strategies and impact on bioprocess design,” Trends Biotechnol., vol. 27, no. 8, pp. 477–485, Aug. 2009.
[4] P. Milburn, J. Bonnerjea, M. Hoare, and P. Dunnill, “Selective flocculation of nucleic acids, lipids, and colloidal particles from a yeast cell homogenate by polyethyleneimine, and its scale-up,” Enzyme Microb. Technol., vol. 12, no. 7, pp. 527–532, Jul. 1990.
[5] I. M. Helander, H. L. Alakomi, K. Latva-Kala, and P. Koski, Microbiology, vol. 143, no. 10, pp. 3193–3199, 1997.
[6] I. M. Helander, K. Latva-Kala, and K. Lounatmaa, Microbiology, vol. 144, no. 2, pp. 385–390, 1998.
